GMPTEC Products
GMPTEC Products
GMPTEC
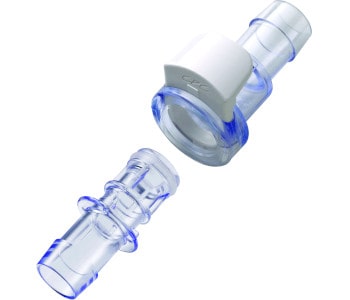
The GMPTEC product portfolio includes a huge variety of components and systems for the production of pharmaceuticals, also diagnostics, chemical products and high-quality foods. Most of the GMPTEC technologies are being used in the biotechnology and both, our single-use and multi-use components, are suitable for cGMP conform biotech-applications. GMPTECs primary goal is to cover all requirements out of one source! This does not only include a huge range of different products, but also services like consulting, assessment and troubleshooting, as well as development and construction.
In terms of customized assemblies, we will gladly take care of the whole product development process! From the first draft, to a technical drawing, to a competitive offer – completely free of charge! Our cleanroom services allow us to create another added value for your processes! We do not only deliver off-the-shelf products in big standardized packaging units: To meet your individual requirements we can clean single GMPTEC goods, channel them into the ISO class 7 cleanroom, also label or laser engrave them, repackage them individually and deliver them directly to you. Right from the start, we make your solutions and interests to our first priority!
We are always available by phone or e-mail and willing to schedule meetings on site even at short notice. The core competence of our team is their wide technical knowledge about the production technologies on the market. We do not settle for only few key products. Instead we want to offer seamless solutions, even if it should be beyond our own portfolio.
GMPTEC
The GMPTEC product portfolio includes a huge variety of components and systems for the production of pharmaceuticals, also diagnostics, chemical products and high-quality foods. Most of the GMPTEC technologies are being used in the biotechnology and both, our single-use and multi-use components, are suitable for cGMP conform biotech-applications. GMPTECs primary goal is to cover all requirements out of one source! This does not only include a huge range of different products, but also services like consulting, assessment and troubleshooting, as well as development and construction.
In terms of customized assemblies, we will gladly take care of the whole product development process! From the first draft, to a technical drawing, to a competitive offer – completely free of charge! Our cleanroom services allow us to create another added value for your processes! We do not only deliver off-the-shelf products in big standardized packaging units: To meet your individual requirements we can clean single GMPTEC goods, channel them into the ISO class 7 cleanroom, also label or laser engrave them, repackage them individually and deliver them directly to you. Right from the start, we make your solutions and interests to our first priority!
We are always available by phone or e-mail and willing to schedule meetings on site even at short notice. The core competence of our team is their wide technical knowledge about the production technologies on the market. We do not settle for only few key products. Instead we want to offer seamless solutions, even if it should be beyond our own portfolio.
NEWS
2024 calendar available
Our new 2024 calendar is now available!
If you’re as excited about it as we are, send a request to info@gmptec.de and we’ll be happy to send you one!
Christmas Party
🎄It was an amazing Christmas Party this year🎄
The first #GMPTEC Winter Games were well received and celebrated! We are proud to be such great and united Team!
Global Bioprocessing Summit in Berlin
Global Bioprocessing Summit in Berlin just started!
GMPTEC GmbH is proud to be part of this event as a sponsor! Meet our colleagues Marco John and Marcel Dombrowsky at our booth and discuss your individual single-use challenges and solutions.
Single-Use Systeme 2023 in Heidelberg
Single-Use Systeme 2023 in Heidelberg!
The first day of this exciting seminar started and we are happy to be part of it!
Visit the GMPTEC GmbH booth and talk to our colleagues Marco John and Marcel Dombrowsky about the latest single-use innovations!
Also don’t miss out our friends Katja Wilms and Michael Schulz from GEA Group right next to us!
BIO-Europe 2023
The BIO-Europe 2023 in Munich has ended yesterday and we are pleased that we were able to support our partner Entegris at the show.
Visit our Partner Savillex
Yesterday, we‘ve started our Minneapolis trip and visted our Partner Savillex ! Thanks to the whole Savillex-Team for a great tour through your facilities and fascinating Impressions!